PCAM Seminar 11. April 2024
Time: 16:15-17:00
We welcome two speakers this time.
Jinan Hussein Awadh Alshuhaib, Universidad Autónoma de Madrid
Sensitized near-infrared lanthanide emission in semiconductor chalcogenide perovskites
Nowadays there is a great interest in using semiconductors as host materials for photoluminescent lanthanide ions (Ln3+). In such a material, the poor light absorption intrinsically featured by lanthanides is compensated by the semiconductor moiety, which harvests the optical energy and funnel it to the luminescent metal center [1]. However, incorporating Ln3+ in semiconductors is difficult to achieve because Ln3+ prefer to occupy sites in the crystal lattice with high coordination number, such as octahedral sites [2], whereas most semiconductors offer cationic sites with tetrahedral coordination.
In this vein, inorganic chalcogenide perovskites can offer a solution. These materials are semiconductors with general formula ABX3, with A being a group II cation (i.e., Ca2+, Sr2+, or Ba2+), B a group IV transition metal (i.e., Ti4+, Zr4+, or Hf4+), and X a chalcogen anion (S2− or Se2−) [3]. The B site features an octahedral coordination environment ideal for embedding Ln3+.
Moving from these considerations, here we present the synthesis and characterization of BaZrS3 perovskites doped with Ln3+ (Yb+3, Er+3, Nd+3). Samples were prepared by sulfurization of Ln3+-doped BaZrO3 powders, which were in turn prepared via a microwave-assisted hydrothermal method. The morphology, chemical composition and crystalline structure of the samples were characterized by scanning electron microscopy coupled to energy dispersive x-ray analysis, Raman spectroscopy and x-ray powder diffraction (XRPD). Optical band gaps were determined by diffuse optical reflectance measurements. Preliminary photoluminescence studies confirm the occurrence of effective energy transfer from the semiconductor host material to the doped Ln3+.
[1] R. Marin, D. Jaque, Chem. Rev. 121, 1425−1462 (2021)
[2] W. Mir et al., NPG Asia Mater 12, 9 (2020)
[3] Y. Sun et al., Nano Letters, 15, 581−585 (2015)
Leyre Aldaz Caballero, Universidad Autónoma de Madrid
Unveiling the luminescence mechanism of γ-In2S3: the effect of incorporation of dopants
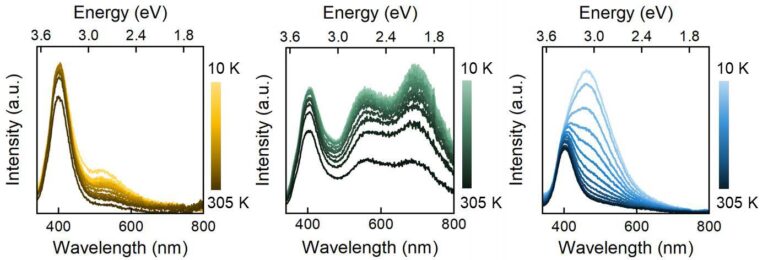
Trivalent lanthanide ions (Ln3+) and transition metal ions (such as Cr3+) are well known for their unique luminescence properties, having narrow emission lines from the visible (Vis) to the near-infrared (NIR) spectral range and long lifetimes (from μs to ms).1, 2 The shortcoming of these ions comes from their low absorption coefficient (0.1-10 M-1 cm-1), and thus, low brightness. This low absorption and emission efficiency is coming the nature of the transitions that they have, which are parity-forbidden. An attractive alternative to address this problem is incorporating these ions with semiconductor nanocrystals, which are small crystalline particles with a high molar absorption coefficient (104-106 M-1 cm-1) and strong emission. By doping SNCs with Ln3+ or Cr3+ ions, we can solve the shortcoming that they present: the SNCs would act as host material, absorbing the incident photons and transfer them to the dopant ion. This process is known as sensitized luminescence. To achieve the incorporation of luminescence ions, γ-indium sulfide (γ-In2S3) was chosen as a host material.3 In this crystal structure, the In3+ ion occupies an octahedral crystallographic site, has the same oxidation state as the dopants (Ln3+ and Cr3+) and has a similar ionic radius. These conditions facilitate both a straightforward incorporation of the dopants into the host material and the likeliness of achieving energy transfer to the ions through excitation of the host material. In this work, we synthesized γ-In2S3 nanoplatelets and dope them with Nd3+, Yb3+ and Cr3+ ions to study the effect that the incorporation generates into the host material and explore if the energy transfer process is possible. This study lays the foundation for future improvements in the design of these materials, by way of unveiling the luminescence mechanism of γ-In2S3 and which energy states are involved.
Figure 1. Low-temperature emission spectra of γ-In2S3, γ-In2S3:Cr3+(5%) and γ-In2S3:Nd3+(5%), λex=260 nm. The emission intensity is quenched while the temperature is increased. The incorporation of dopants changes the emission shape from the samples.
References.
1. Marin, R.; Jaque, D., Doping Lanthanide Ions in Colloidal Semiconductor Nanocrystals for Brighter Photoluminescence. Chemical Reviews 2021, 121 (3), 1425-1462.
2. Dang, P.; Wei, Y.; Liu, D.; Li, G.; Lin, J., Recent Advances in Chromium-Doped Near-Infrared Luminescent Materials: Fundamentals, Optimization Strategies, and Applications. 2023, 11 (3), 2201739.
3. Park, K. H.; Jang, K.; Son, S. U. J. A. C. I. E., Synthesis, Optical Properties, and Self‐Assembly of Ultrathin Hexagonal In2S3 Nanoplates. 2006, 45 (28), 4608-4612.
